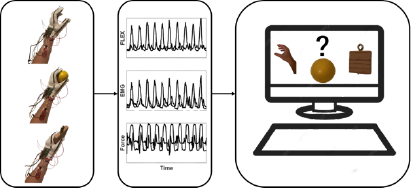
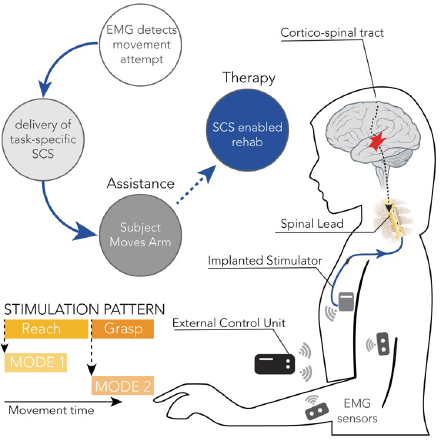
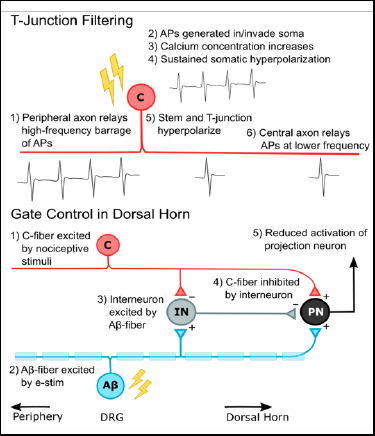
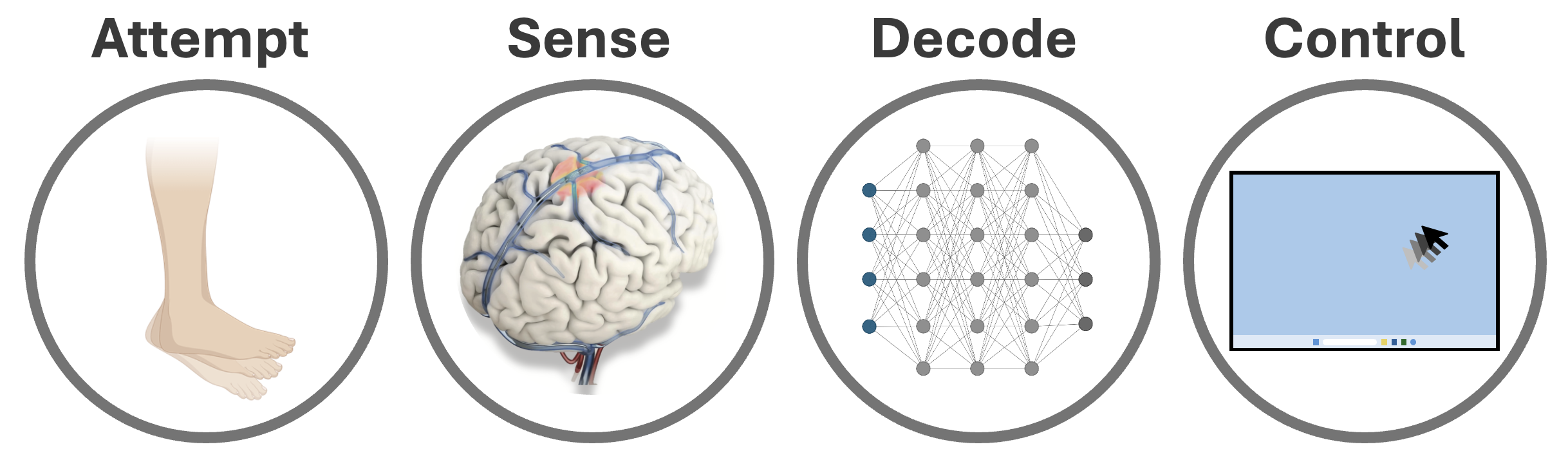
NeuroMechatronics Lab
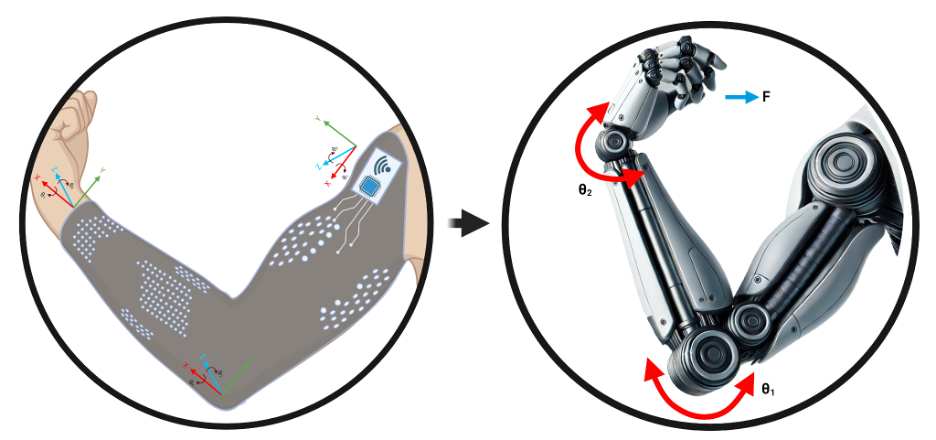
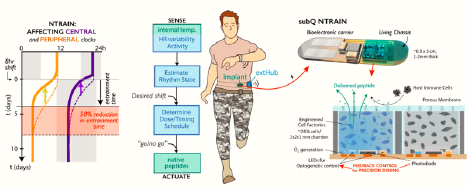
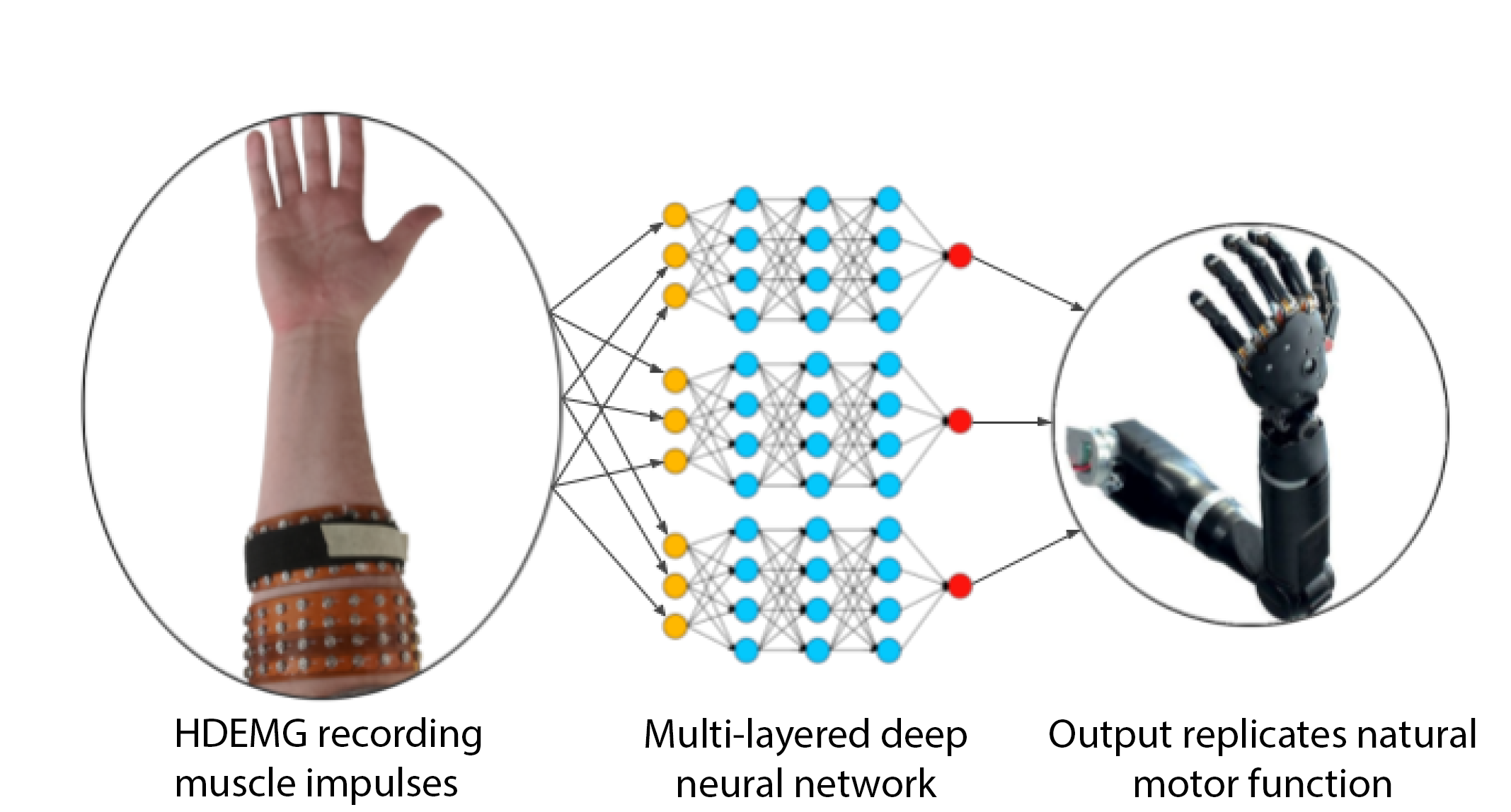
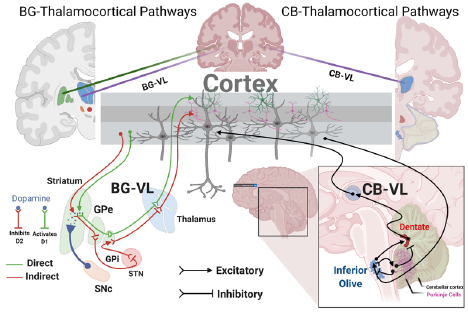
The NeuroMechatronics lab is home to an inclusive and multidisciplinary community of neuroscientists and engineers. We work collaboratively to understand the neurophysiology of sensory and motor systems in the body. We strive to invent new strategies and devices that will allow us to transform the treatment of neurological disorders. Our research spans the full spectrum from fundamental to applied research and we leverage an expansive network of academic and industry partners to tackle important problems aggressively and at scale. Throughout it all, we focus on the neuroethical underpinnings and wider societal implications of our neurotechnology.